
Dr. Reddy’s Laboratories announced that it has launched Sumatriptan Injection USP, Autoinjector System 6 mg/0.5 mL, for Subcutaneous Use, a therapeutic equivalent generic version of IMITREX STATdose Pen® (sumatriptan succinate) 6 mg/0.5 mL in the US market on Feb. 25, 2014, following the approval by the United States Food & Drug Administration (USFDA).
 The IMITREX STATdose Pen® (sumatriptan succinate) brand and generic combined had U.S. sales of approximately $169 million MAT for the most recent twelve months ending in December 2013 according to IMS Health*.
The IMITREX STATdose Pen® (sumatriptan succinate) brand and generic combined had U.S. sales of approximately $169 million MAT for the most recent twelve months ending in December 2013 according to IMS Health*.
Dr. Reddy's Sumatriptan Injection USP, Autoinjector System 6 mg/ 0.5 mL is available in a carton containing 2 single-dose prefilled syringes.
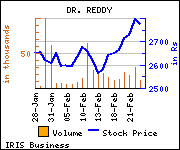
Shares of the company declined Rs 21.25, or 0.76%, to settle at Rs 2,773.90. The total volume of shares traded was 11,258 at the BSE (Tuesday).