 Strides Arcolab, a pharmaceutical company manufacturing IP-led niche products, witnessed a rise in share price on Friday after it has received approval from the United States Food & Drug Administration (USFDA) for Lamivudine and Zidovudine tablets USP, 150 mg/ 300 mg.
Strides Arcolab, a pharmaceutical company manufacturing IP-led niche products, witnessed a rise in share price on Friday after it has received approval from the United States Food & Drug Administration (USFDA) for Lamivudine and Zidovudine tablets USP, 150 mg/ 300 mg.
 According to IMS data, the US market for Lamivudine and Zidovudine tablets is approximately USD 120 million. The product will be manufactured at the company's oral dosage facility at Bangalore and marked by Strides in the US Market. The product will be launched in the markets immediately.
According to IMS data, the US market for Lamivudine and Zidovudine tablets is approximately USD 120 million. The product will be manufactured at the company's oral dosage facility at Bangalore and marked by Strides in the US Market. The product will be launched in the markets immediately.
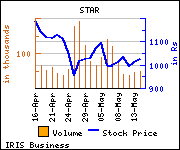
Shares of the company are trading at Rs 1,068.55, up Rs 44.5, or 4.35% at the Bombay Stock Exchange (BSE) on Friday at 11:42 a.m.
Total volume of shares traded on the bourses today was higher by 193.27% to 489,046 compared with 22-day average volume of 166,756.