Lupin plunges 4% on concerns over Suprax sales
Source: IRIS | 16 Apr, 2015, 01.24PM
|
|
|
|
|
| Rating: NAN / 5 stars. |
 Comments | Post Comment Comments | Post Comment |
|
 Lupin, one of the leading pharmaceutical companies in India, witnessed a sharp decline in share price on Thursday concerns over sales of Suprax after another drug maker received approval from US drug regulator for the same product. Lupin, one of the leading pharmaceutical companies in India, witnessed a sharp decline in share price on Thursday concerns over sales of Suprax after another drug maker received approval from US drug regulator for the same product.
Aurobindo Pharma got a nod from US Food and Drug Administration (USFDA) to make and sell its generic version of oral suspension of antibiotic Cefixime. The approved ANDAs are bioequivalent and therapeutically equivalent to the reference listed drug product (RLD) Suprax® Oral Suspension USP 100mg/5mL and 200mg/5mL respectively of Lupin Pharmaceuticals Inc.
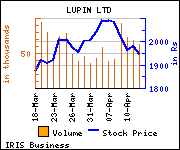 Shares of the company are trading at Rs 1,869.70, down Rs 77.9, or 4% at the Bombay Stock Exchange (BSE) on Thursday at 1:11 p.m. Shares of the company are trading at Rs 1,869.70, down Rs 77.9, or 4% at the Bombay Stock Exchange (BSE) on Thursday at 1:11 p.m.
The scrip has touched an intra-day high of Rs 1,941.50 and low of Rs 1,868.20. The total volume of shares traded at the BSE is 68,489.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|