IOL Chemicals and Pharmaceuticals (IOLCP), a manufacturer and supplier of industrial chemicals, has received United State Food and Drug Administration (US FDA), establishment inspection report after successful inspection of its Ibuprofen manufacturing facility situated at Barnala, Punjab, India.
 Post this approval, IOLCP will garner additional business for its flagship API Ibuprofen in world's largest pharma market-USA. This will result into higher value added export turnover and higher margins.
Post this approval, IOLCP will garner additional business for its flagship API Ibuprofen in world's largest pharma market-USA. This will result into higher value added export turnover and higher margins.
The company's manufacturing facility already holds CEP certification for its products Ibuprofen. Metformin Hydrochloride & Lamotrigine from European Directorate for the Quality of Medicines & HealthCare (EDQM) authorities, council of Europe, France.
The Barnala facility has also been approved by ANVISA Brazil, WHO & MOH Japan.
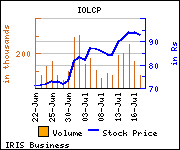
Shares of the company gained Rs 13.7, or 14.76%, to trade at Rs 106.50. The total volume of shares traded was 488,660 at the BSE (2.47 p.m., Monday).