 Ranbaxy Labs has received the regulatory approval to launch India's first New Chemical Entity (NCE), Synriam (arterolane maleate 150 mg + piperaquine phosphate 750 mg drug) in seven African countries, namely, Nigeria, Uganda, Senegal, Cameroon, Guinea, Kenya and Ivory Coast. The company said the product had been launched in Uganda and would be made available in other countries towards end of January 2015.
Ranbaxy Labs has received the regulatory approval to launch India's first New Chemical Entity (NCE), Synriam (arterolane maleate 150 mg + piperaquine phosphate 750 mg drug) in seven African countries, namely, Nigeria, Uganda, Senegal, Cameroon, Guinea, Kenya and Ivory Coast. The company said the product had been launched in Uganda and would be made available in other countries towards end of January 2015.
 The new drug conform to the recommendations of the World Health Organization (WHO) for using combination therapy in malaria. Synriam provides quick relief from most malaria-related symptoms, including fever, and has a high cure rate of over 95 percent. Ranbaxy is also conducting Phase III clinical trials for the pediatric formulation of Synriam in pediatric patients of uncomplicated Plasmodium falciparum malaria.
The new drug conform to the recommendations of the World Health Organization (WHO) for using combination therapy in malaria. Synriam provides quick relief from most malaria-related symptoms, including fever, and has a high cure rate of over 95 percent. Ranbaxy is also conducting Phase III clinical trials for the pediatric formulation of Synriam in pediatric patients of uncomplicated Plasmodium falciparum malaria.
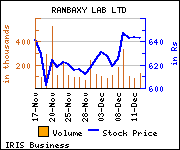
Commenting on the same, Sarabjit Kour Nangra, VP research, pharma, Angel Broking, said, ''A rough estimate puts the market for the drug in these markets at around USD 300-350 million, on a conservative basis. Thus the development is positive and can add around easily around USD 80-100 million, within first year of sales and would be fully reflected in FY2016. We remain our neutral rating on the stock.''