Ajanta Pharma, a company engaged in manufacturing & marketing of pharmaceutical products on Monday said its subsidiary Ajanta Pharma USA Inc announced the launch of Montelukast Sodium Oral Granules (4mg), a bioequivalent generic version of Singulair Granules, in the US market. Ajanta Pharma offers Montelukast Granules in cartons containing 30 individual sachets.
 The approval and launch of the Montelukast Granules is part of an ever-growing portfolio that Ajanta Pharma has developed for the US market.
The approval and launch of the Montelukast Granules is part of an ever-growing portfolio that Ajanta Pharma has developed for the US market.
To date, the United States Food & Drug Administration (US FDA) has granted five ANDA final approvals and two ANDA tentative approvals. An additional 19 ANDAs are pending approval from the US FDA, the company said.
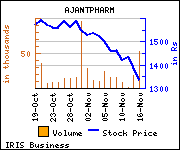
Shares of the company declined Rs 55.85, or 4.02%, to settle at Rs 1,334.55. The total volume of shares traded was 53,195 at the BSE (Monday).