 Lupin, one of the leading pharmaceutical companies in India, announced Thursday that it has received final approval for its Omeprazole delayed-release capsules 40 mg from the United States Food and Drugs Administration (USFDA).
Lupin, one of the leading pharmaceutical companies in India, announced Thursday that it has received final approval for its Omeprazole delayed-release capsules 40 mg from the United States Food and Drugs Administration (USFDA).
 The company got USFDA approval to market a generic version of AstraZeneca Pharmaceuticals LP Prilosec delayed-release capsules 40 mg.
The company got USFDA approval to market a generic version of AstraZeneca Pharmaceuticals LP Prilosec delayed-release capsules 40 mg.
Lupin's Omeprazole delayed-release capsules 40 mg are the AB rated generic equivalent of AstraZeneca's Prilosec delayed-release capsules 40 mg.
It is indicated for the short-term treatment of Duodenal Ulcer, Gastric Ulcer, treatment of Gastro Esophageal Reflux Disease (GERD), maintenance of healing of Erosive Esophagitis and Pathological Hypersecretory conditions.
According to IMS MAT June 2015, Prilosec had annual sales of USD 185 million.
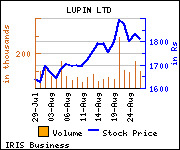
Shares of the company gained Rs 86.7, or 4.79%, to settle at Rs 1,898.50. The total volume of shares traded was 293,958 at the BSE (Thursday).